CLINICAL DEVELOPMENT & RESEARCH
The results reported here concern the studies with the portable J-PET prototype constructed at the Jagiellonian University in Poland and conducted by the researchers at the Jagiellonian University.
So far two registered studies were conducted:
1) „Clinical Application of the Prototype J-PET Device (JPETClinic)”
https://clinicaltrials.gov/study/NCT06211803
ClinicalTrials.gov ID NCT06211803
Conduced at the Medical University of Warsaw, Poland
2) „Clinical Application of the J-PET Scanner Prototype (JPET2Clinic)”
https://clinicaltrials.gov/study/NCT06242119
ClinicalTrials.gov ID NCT06242119
Conducted at the University Hospital in Cracow, Poland
Below is a short summary of the preclinical and clinical studies performed by the research group from the Jagiellonian University.
1) Clinical studies at the hospital of the Medical University of Warsaw conducted in March 2022.
Studies registered as:
„Clinical Application of the Prototype J-PET Device (JPETClinic)”
https://clinicaltrials.gov/study/NCT06211803
ClinicalTrials.gov ID NCT06211803
The research was performed at the Medical University of Warsaw, with studies conducted following routine procedures so as not to interfere with routine diagnostics and therapy. If a patient agreed to stay longer on the platform, we had about 10 to 15 minutes to install the J-PET tomograph around them and collect data.
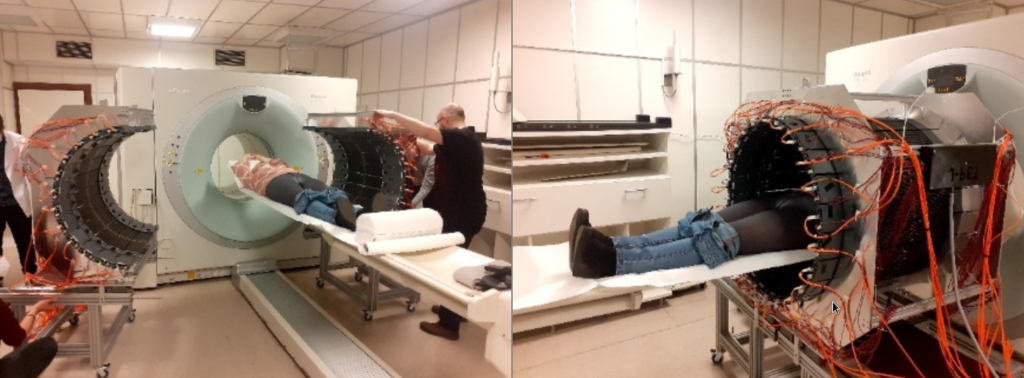
Fig. 2. Photographs of the first imaging of a patient in the hospital of Warsaw Medical University, illustrating the advantages of the J-PET as a portable, lightweight device with an adaptable imaging volume. Images from the public plenary lecture of Prof. Moskal at Japanese Society of Nuclear Medicine: [PL2] Jagiellonian PET: Democratizing access to positron emission tomography with low-cost, modular, lightweight, and portable PET scanners based on plastic scintillators |JSNM2025/JSNMT2025 | Confit
These studies included comparison between J-PET scanner and the Siemens PET/CT Biograph 64 TruePoint by imaging with pharmaceuticals [ 68 Ga]Ga-DOTA-TATE, [ 68 Ga]Ga-PSMA-11, [68Ga]Ga-DOTA-SP and therapy with [225Ac]Ac-DOTA-SP.
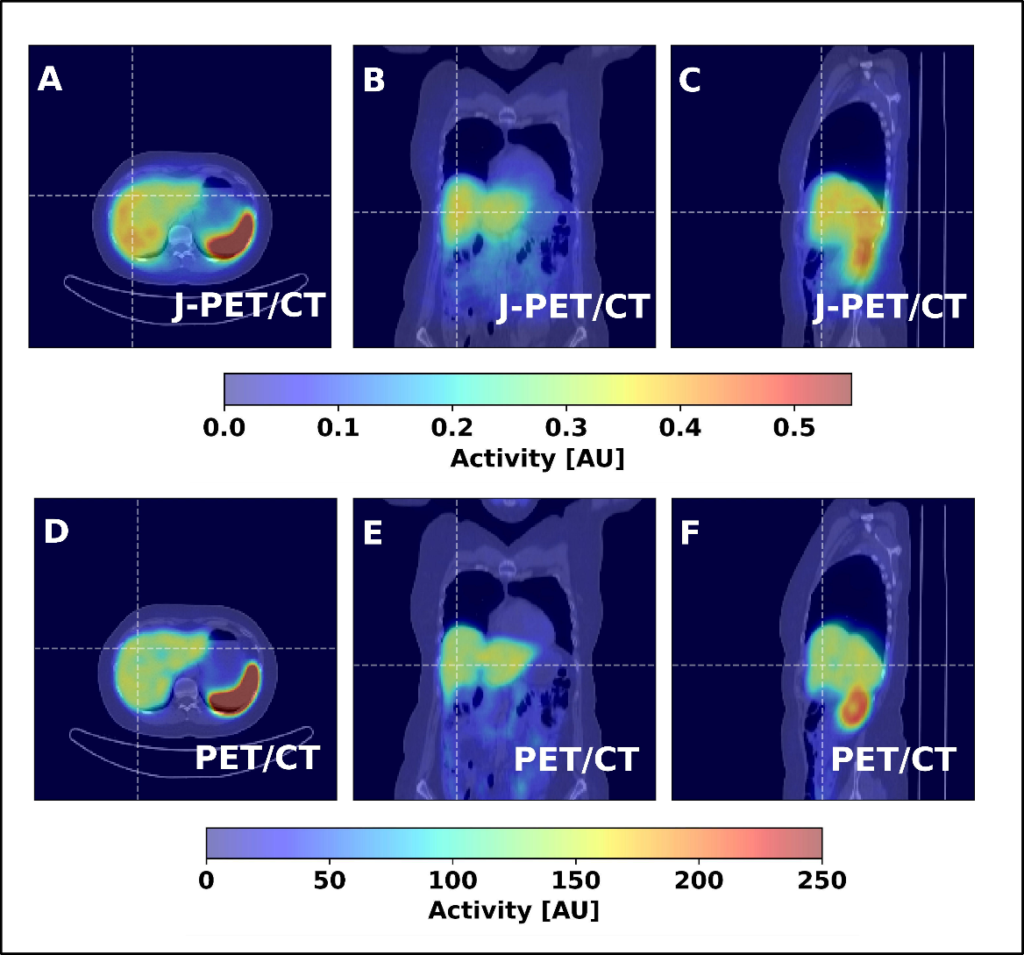
Fig. 3. Coronal, sagittal, and transversal images obtained from a patient injected with the [68Ga]Ga-DOTA-TATE pharmaceutical, comparing a 23-minute scan with a Siemens PET/CT to a 15-minute scan with a J-PET. Images from the public plenary lecture of Prof. Moskal at Japanese Society of Nuclear Medicine: [PL2] Jagiellonian PET: Democratizing access to positron emission tomography with low-cost, modular, lightweight, and portable PET scanners based on plastic scintillators |JSNM2025/JSNMT2025 | Confit
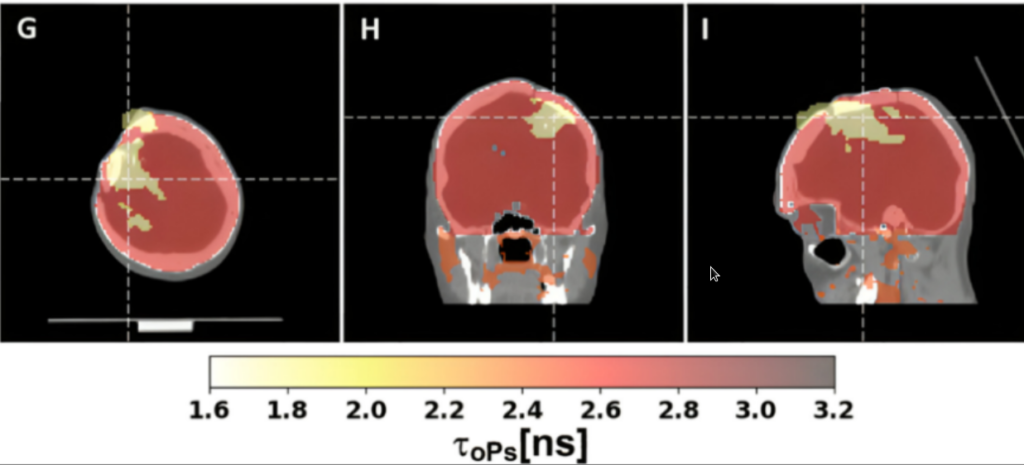
Fig. 4. This is the first ever positronium image of the human brain which was demonstrated using the J-PET scanner. Positronium images of a patient with glioblastoma, showing the difference in mean ortho-positronium lifetime (a new diagnostic parameter) between glioma and healthy brain. The figure is taken from the article published in Science Advances: https://www.science.org/doi/10.1126/sciadv.adp2840
Fig 4. presents first ever positronium image of the human brain performed using the method patented by the J-PET. It shows results of theranostics studies using pharmaceuticals [68Ga]Ga-PSMA-11 and [68Ga]Ga-DOTA-SP for imaging and [225Ac]Ac-DOTA-SP for therapy of glioma cancer.
The first patient for positronium imaging was a 45-year-old man with glioblastoma (an aggressive brain tumour) undergoing alpha-particle radiotherapy. The primary aim of his therapy was to destroy the tumour using alpha particles emitted by the radionuclide 225 Ac. The positronium imaging was made possible by the concurrent theranostic application of the radionuclide 68 Ga to monitor the site of cancer lesions using a PET scanner.
The patient was administered a simultaneous intra-tumoural injection of the alpha-particle-emitting radiopharmaceutical ( 225 Ac-DOTA-SP) for therapy and the positron emitting pharmaceutical ( 68 Ga-DOTA-SP) for diagnosis. In about 1% of cases, after emitting a positron that annihilates with an electron, 68 Ga also emits a prompt gamma ray.
We determined the annihilation location by measuring the time and position of interaction of the annihilation photons in the scanner. For each image voxel, we also determined a lifetime spectrum as the distribution of differences between the time of annihilation and the time of prompt gamma emission.
Our study found that positronium lifetimes in glioblastoma cells are shorter than in salivary glands and healthy brain tissues. We showed for the first time that the mean lifetime of ortho-positronium in a glioma (1.77±0.58 ns) is shorter than in healthy brain tissue (2.72±0.72 ns). This finding demonstrates that positronium imaging could be used for vivo diagnosis to differentiate between healthy and cancerous tissues.
The result shown above is published in the journal Science Advances
https://www.science.org/doi/10.1126/sciadv.adp2840
and it was also described by Science
https://www.science.org/content/article/new-twist-pet-scans-unlocks-hidden-signals-diagnosing-disease
and by Physics World
https://physicsworld.com/a/towards-quantum-pet-harnessing-the-diagnostic-power-of-positronium-imaging/
2) Clinical studies at the University Hospital in Cracow conducted in March 2024
Studies registered as:
„Clinical Application of the J-PET Scanner Prototype (JPET2Clinic)”
https://clinicaltrials.gov/study/NCT06242119
ClinicalTrials.gov ID NCT06242119
Studies included comparison between J-PET scanner and the GE Discovery MI Gen 2 PET/CT by imaging patients with pharmaceuticals: [ 68 Ga]Ga-DOTA-TATE, [ 68 Ga]Ga-PSMA, and [ 18 F]FDG.
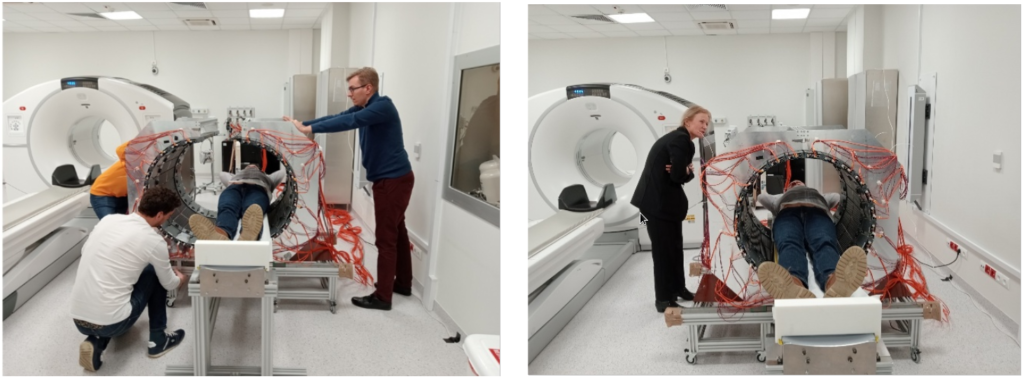
Fig. 5. Photographs of the first imaging of a patient in the University Hospital in Cracow, illustrating the advantages of the J-PET as a portable, lightweight device with an adaptable imaging volume.
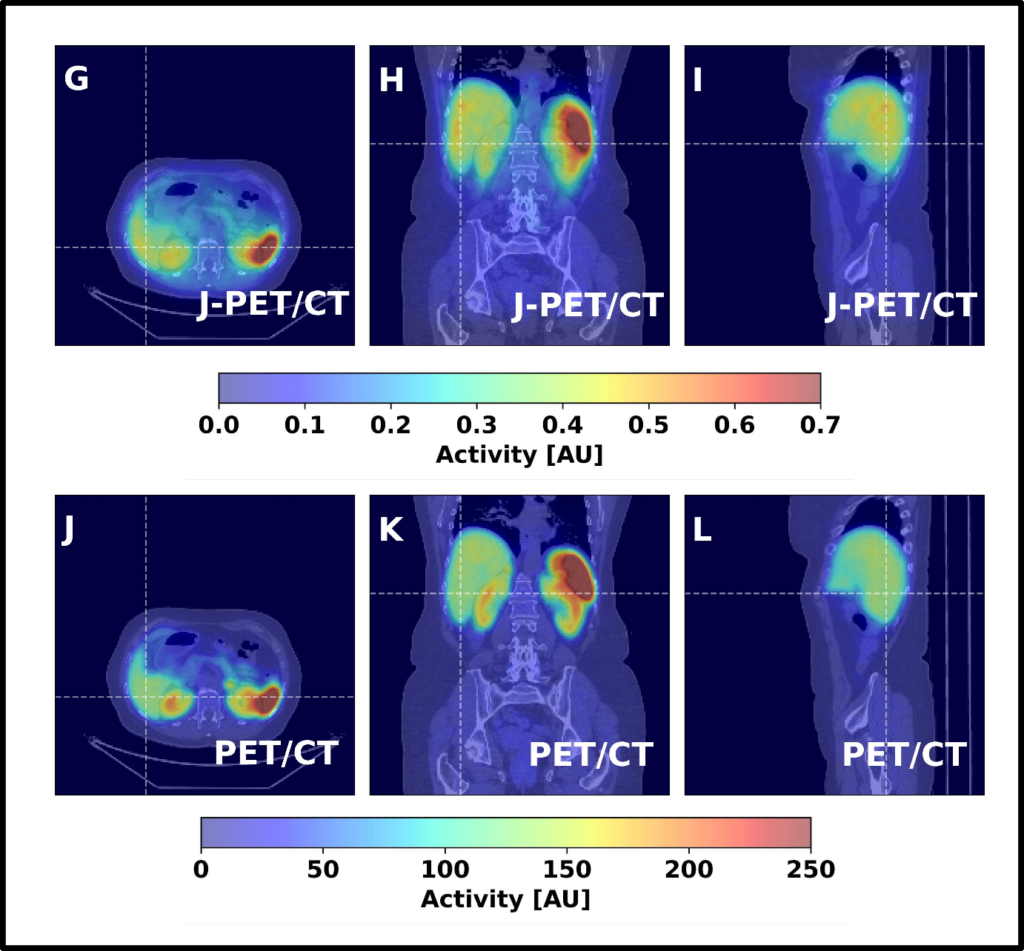
Fig. 6. Coronal, sagittal, and transversal images obtained from a patient injected with the [68Ga]Ga-DOTA-TATE pharmaceutical, comparing a 25-minute scan with GE Discovery MI Gen 2 PET/CT to a 16-minute scan with a J-PET. Images are from the public plenary lecture of Prof. Moskal at Japanese Society of Nuclear Medicine: [PL2] Jagiellonian PET: Democratizing access to positron emission tomography with low-cost, modular, lightweight, and portable PET scanners based on plastic scintillators | JSNM2025/JSNMT2025 | Confit
3) The above shown clinical studies were preceded by preclinical studies at the Proton Therapy Center in Cracow conducted by the research group from the Jagiellonian University in October 2021.
The studies were conducted in the Proton Therapy Center in Cracow. They have proven the capability of the J-PET scanner for application of proton beam therapy monitoring.

Fig. 2. Photographs taken in the Proton Therapy Center in Cracow showing the first therapeutic proton beam monitoring using J-PET modular scanner arranged in the two sets of three layers. In the center of right photograph, between the modules an image of the proton beam obtained with J-PET is shown. The proton beam was irradiating the phantom from above. An image taken with J-PET clearly shows the range of the irradiation. The achieved precision of the determination of the range of proton beam is better than 1mm.
Images are from the public plenary lecture of Prof. Moskal at Japanese Society of Nuclear Medicine: [PL2] Jagiellonian PET: Democratizing access to positron emission tomography with low-cost, modular, lightweight, and portable PET scanners based on plastic scintillators | JSNM2025/JSNMT2025 | Confit
